17.04.2026
Industries: Health, Life Sciences & Pharmaceuticals
EUDAMED: Mandatory Modules Effective May 2026
On November 26, 2025, the European Commission published Decision (EU) 2025/2371 (“Decision”), confirming the full operationality of the first four modules of the European Database on Medical Devices (EUDAMED).
Its use will become mandatory as of May 28, 2026, and is relevant for economic operators subject to Regulation (EU) 2017/745 (“MDR”) and Regulation (EU) 2017/746 (“IVDR”).
What is EUDAMED?
EUDAMED is the European database on medical devices, created with the aim of centralizing and making available information regarding all aspects of the life cycle of medical devices in the European Union. When fully operational, EUDAMED will consist of six modules.
Which Modules have been declared operational?
Following an independent audit completed on June 18, 2025, the Commission confirmed that the following electronic systems are operational:
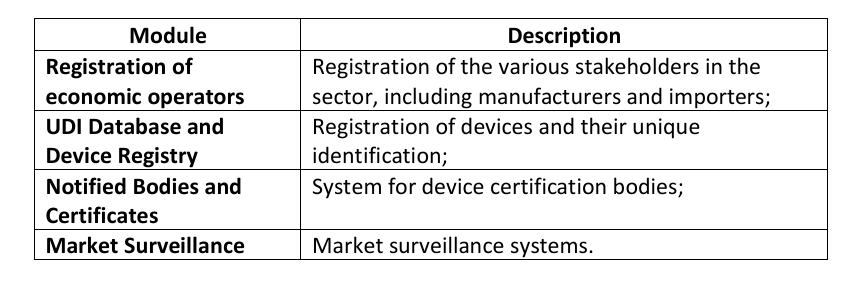
Among the identified benefits are the provision of a single registration point across the EU, the assignment of a Unique Registration Number (“URN”), and the potential reduction of duplicative bureaucratic obligations.
Impact on Portuguese Registrations and Next Steps
Although the Medical Device Information System (“SIDM”) is currently in force, with EUDAMED becoming mandatory as of May 28, 2026, the national registration obligations may be affected, and it will be necessary to await guidance from INFARMED to determine the specific impact on the requirements applicable to each category of economic operator.
Therefore, economic operators are advised to:
- Check the status of their registrations in EUDAMED and assess any pending registrations or notifications;
- monitor the guidance to be issued by INFARMED; and
- review their internal compliance processes in light of the applicable obligations.



























